 |
Centre for Australian National Biodiversity
Research
|
Supervisor: Mark Clements
 Oligochaetochilus gibbosus, the Illawarra Greenhood Orchid, is an endangered
orchid species that is endemic to the NSW central coast. Although protecting
the habitat of this species is an important conservation strategy, artificial
propagation is another method that could be used to help conserve O. gibbosus.
This would involve germinating seed collected from wild plants, and then the
translocation of seedlings back into their natural habitat.
Oligochaetochilus gibbosus, the Illawarra Greenhood Orchid, is an endangered
orchid species that is endemic to the NSW central coast. Although protecting
the habitat of this species is an important conservation strategy, artificial
propagation is another method that could be used to help conserve O. gibbosus.
This would involve germinating seed collected from wild plants, and then the
translocation of seedlings back into their natural habitat.
In the wild, all orchids require a relationship with a mycorrhizal fungus in order for their seed to germinate. This relationship is often used when artificially propagating orchids from seed (the technique is known as symbiotic seed germination) in order to create seedlings with a higher chance of survival than those grown through asymbiotic methods.
When thinking about how to do this, several questions arise. If the seed is to be symbiotically germinated, which fungus will be most successful in terms of germination rate? Also, there is some O. gibbosus seed that has been in long-term storage for up to 17 years, including some that was collected from an area where O. gibbosus is now virtually extinct. It is important that the viability of this seed, and hence its potential to contribute to the conservation of O. gibbosus is determined, particularly with relation to preserving the genetic diversity of the species, a vital factor when attempting to conserve this orchid.
These questions lead to two hypotheses that were tested in several experiments
1. There are no differences between the effects of different mycorrhizal fungal isolates in symbiotic seed germination of O. gibbosus?
2. There are no differences in seed germination of O. gibbosus when using stored seed of varying ages.
The purpose of this experiment was to determine the effectiveness of various mycorrhizal fungi. Hence, 6 fungal isolates were chosen: 2 from O. gibbosus, 2 from Hymenochilus bicolor (a closely related genus to Oligochaetochilus, collected from sites where O. gibbosus was present), 1 from O. rufus (collected from sites where O. gibbosus was not present) and 1 from O. leptochilus (an orchid from Western Australia).
For this experiment, the variable was seed, with seeds stored for different amounts of time being used. The seed was originally collected in: 1997 (stored at room temperature), 1985, 1982, 1981, 1980, 1978 and 1976.
For both experiments the method was basically the same:
Data was taken by scoring seeds present in a small sample area according to the germination stage they were at, using a scale of 0 to 6 (where 0 is dry seed, no germination, and 6 is a seedling ready to be transferred to soil). Due to the short period that the experiments had been running for, the most developed protocorms had only reached germination stage 2 (breaking the seedcoat).
Experiment 1
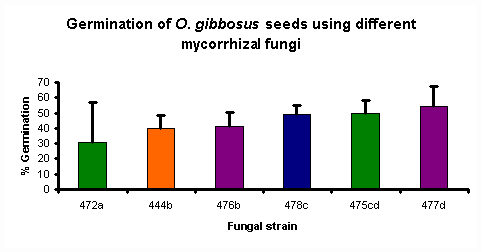
Figure 1
Figure 1 shows the percentage germination of O. gibbosus seeds obtained using the various fungal isolates. Statistical analysis of the results shows that 4 groups of fungi can be formed based on their effectiveness, as shown by the letters a, b, c, d.
It appears that the fungal isolate used will have a definite effect on the percentage germination obtained. However, this effect cannot be linked to species or location of the orchid that the fungus was isolated from. This makes it difficult to predict, when only knowing these factors, the extent to which a particular isolate will enhance orchid seed germination without actually testing it and observing the result.
Experiment 2
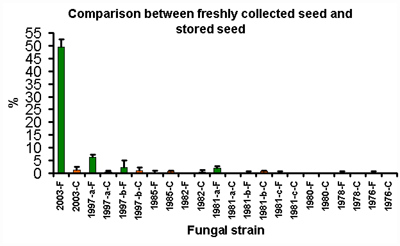
Figure 2
Figure 2 shows the percentage germination obtained using seeds stored for various periods of time. The extremely low germination rates that are present in seed that has been in long term storage (at best, about 6% in comparison to about 50% for the freshly collected seed) indicate that orchid seed does not have the capacity to survive storage in the manner which has occurred in the herbarium. A regression analysis was performed on the data, and the results are shown in figure 3 below.
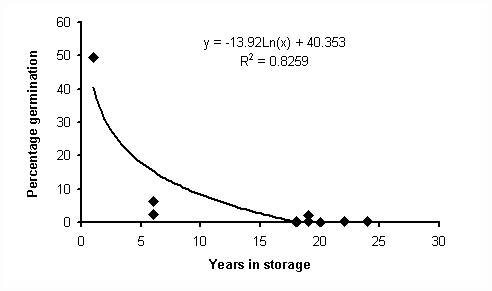
Figure 3
The regression analysis demonstrates that there is an exponential decay in percentage germination over time. However, there is the need for further experimentation to determine the exact shape of the curve (by filling in data gaps, especially between 1 and 5 year old seed).